Williams
Laboratory
Mechanisms and Modulators of Wnt Signaling in Development and Disease
 Wnt signaling is a fundamental regulator of embryonic development, tissue homeostasis and regeneration. Disruption of this pathway contributes to a wide spectrum of human diseases, including congenital skeletal disorders and aggressive cancers.
Wnt signaling is a fundamental regulator of embryonic development, tissue homeostasis and regeneration. Disruption of this pathway contributes to a wide spectrum of human diseases, including congenital skeletal disorders and aggressive cancers.
Wnt ligands signal through Frizzled (Fzd) receptors and associated co-receptors to activate distinct downstream pathways, broadly categorized as canonical (β-catenin–dependent) and noncanonical (β-catenin–independent) signaling. Precise spatial and temporal control of Wnt signaling is essential for normal development and tissue homeostasis, while dysregulation drives disease.
Despite decades of study, fundamental questions remain. How do individual Wnt ligands selectively engage specific Frizzled receptors in vivo? How might receptor conformation and post-translational modifications shape signaling output? And how does cellular context tune pathway activity? In particular, receptor-level mechanisms that diversify Wnt signaling and coordinate canonical and noncanonical pathways remain major gaps in the field.
The Williams Lab investigates how Wnt receptors and downstream signaling networks function in vivo, and how their dysregulation causes developmental disorders and disease. By integrating developmental biology with disease-relevant models, we aim to uncover principles of Wnt signaling that explain how the same pathway can produce distinct outcomes across tissues and disease states.
Our interdisciplinary research program combines mouse genetics, molecular and cell biology, organoid models, and emerging spatial technologies to study Wnt signaling in physiologically relevant contexts. This allows us to link molecular mechanisms at the receptor and signaling complex level to tissue-scale phenotypes and disease progression.
For more information on the specific projects in our lab, visit the “Projects” page ➔
Featured News
Learn More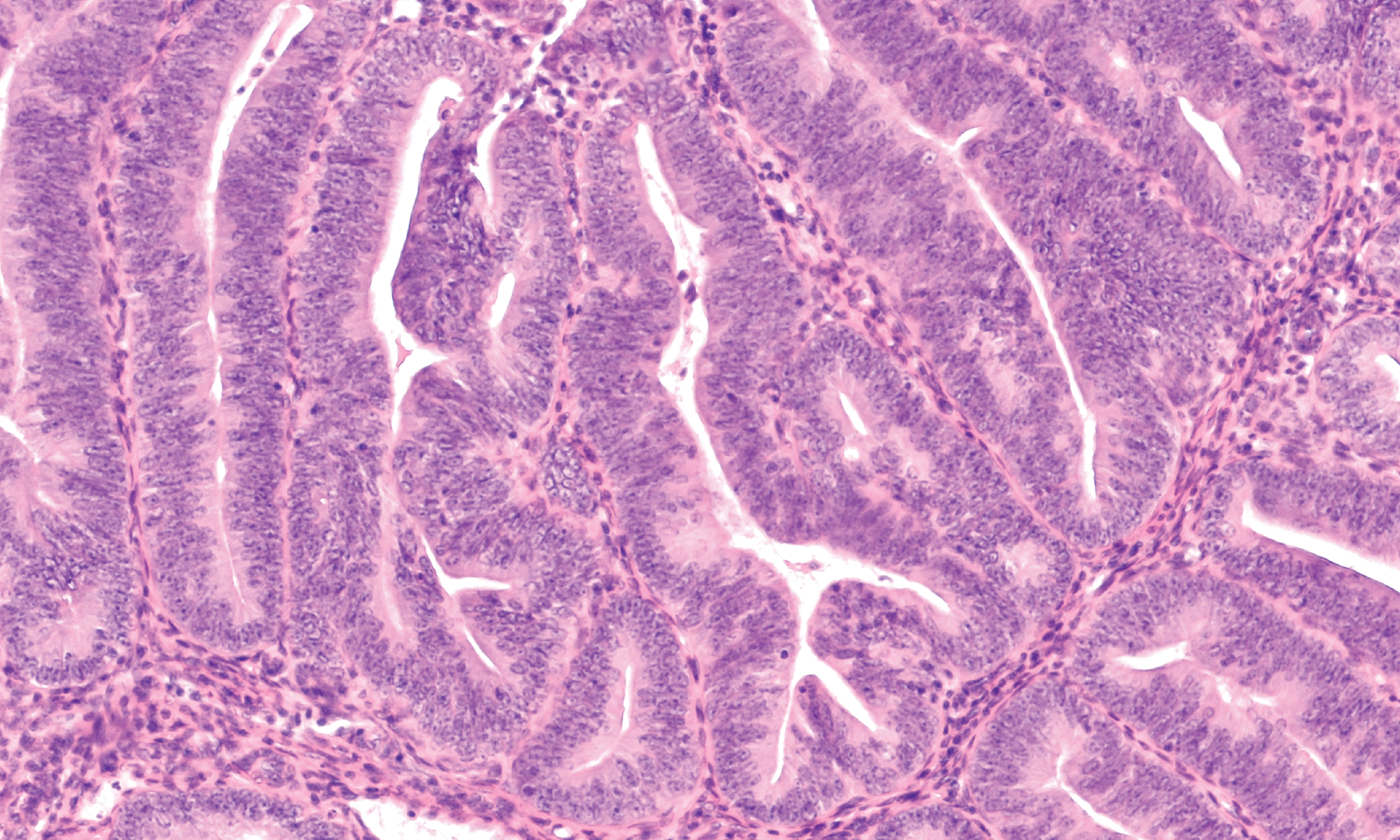
New model offers powerful tool for studying endometrial cancer
What you need to know about sarcoma and bone cancer

Studies do not find hormonal role for bone protein osteocalcin
Diegel CR, Kramer I, Moes C, Foxa GE, McDonald MJ, Madaj ZB, Guth S, Liu J, Harris JL, Kneissel M, Williams BO. 2023. Inhibiting WNT secretion reduces high bone mass caused by Sost loss-of-function or gain-of-function in Lrp5. Bone Res 11(1):47.
Liegel RP, Michalski MN, Vaidya S, Bitterman E, Finnerty E, Menke CA, Diegel CR, Zhong ZA, Williams BO, Stottman RW. 2023. Successful therapeutic intervention in new mouse models of frizzled 2-associated congenital malformations. Development 150(3).
*Featured by the journal
Our Impact
We’re raising thousands to save millions.
We’re turning hope into action for the millions of people around the world affected by diseases like cancer and Parkinson’s. Find out how you can help us make a difference.
- 141 peer-reviewed papers published in 2025, 74 of which were in high-impact journals
- 15 VAI-SU2C Epigenetics Dream Team clinical trials launched to date
- 10 clinical trials and related projects supported by VAI through the International Linked Clinical Trials Program
Bart Williams, Ph.D.
Director, Core Technologies and Services; Professor, Department of Cell Biology
Areas of Expertise
Wnt signaling, skeletal metastases, normal bone development, genetic models, skeletal diseases
Biography
Dr. Williams received his Ph.D. in biology from Massachusetts Institute of Technology in 1996, where he trained with Tyler Jacks. For three years, he was a postdoctoral fellow at the National Institutes of Health in the laboratory of Harold Varmus, former Director of NIH. Dr. Williams joined VAI as a scientific investigator in July 1999. He was promoted to professor in 2013 and served as chair of VAI’s Department of Cell Biology from 2010 to 2022. In 2022, he was appointed director of VAI’s Core Technologies and Services, which provides leading-edge shared scientific resources to bolster research at the Institute and beyond.
2025
Williams BO, Zhong Z, Diegel C, Michalski M, Christie C, Lanning N, Haab B, Grainger S. Preprint. Local misalignment scoring reveals spatially uniform chondrocyte disorganization in a Wnt5a-C83S knock-in model of Robinow syndrome. Bone Res.
Satpathy S … Clinical Proteomic Tumor Analysis Consortium*. 2025. Integrative analysis of lung adenocarcinoma across diverse ethnicities and exposures. Can Cell 42(9):1731–1757.
*Dr. Williams is a member of the Clinical Proteomic Tumor Analysis Consortium
Liu J … Clinical Proteomic Tumor Analysis Consortium*. 2025. Multi-scale signaling and tumor evolution in high-grade gliomas. Can Cell 42(7):1217–1238.
*Dr. Williams is a member of the Clinical Proteomic Tumor Analysis Consortium
Diegel CR, Michalski MN, Ubels JL, Wiartalla GF, Lin CM, Zhong ZA, McDonald MJ, Ethen NJ, Brookshire M, Madaj AB, Xia M, Gavine PR, VAI Transgenic Core, Antonetti DA, Williams BO. 2025. The class A repeats of Lrp5 are required for normal development of bone, retinal vasculature, and mammary gland in vivo. Dis Model Mech.
Yue F, Ku AT, Stevens PD, Michalski MN, Jiang W, Tu J, Shi Z, Dou Y, Wang Y, Feng X-H, Hostetter G, Wu X, Huang S, Shroyer NF, Zhang B, Williams BO, Liu Q, Lin X, Li Y. 2025. Loss of ZNRF3/RNF43 unleashes EGFR in cancer. eLife.
2024
Han D, Labaf M, Zhao Y, Owiredu J, Zhang S, Patel K, Venkataramani K, Steinfeld JS, Han W, Li M, Liu M, Wang Z, Besschetnova A, Patalano S, Mulhearn MJ, Macoska JA, Yuan X, Balk SP, Nelson PS, Plymate SR, Gao S, Siegfried KR, Liu R, Stangis MM, Foxa G, Czernik PJ, Williams BO, Zarringhalam K, Li X, Cai C. 2024. Androgen receptor splice variants drive castration-resistant prostate cancer metastasis by activating distinct transcriptional programs. J Clin Invest 134(11):e168649.
Diegel CR, Michalski MN, Williams BO. 2024. β-catenin-dependent high bone mass induced by loss of APC in osteoblasts does not require Lrp5 or Lrp6. MicroPubl Biol.
2023
Diegel CR, Kramer I, Moes C, Foxa GE, McDonald MJ, Madaj ZB, Guth S, Liu J, Harris JL, Kneissel M, Williams BO. 2023. Inhibiting WNT secretion reduces high bone mass caused by Sost loss-of-function or gain-of-function in Lrp5. Bone Res 11(1):47.
Michalski MN, Williams BO. 2023. The past, present, and future of genetically engineered mouse models for skeletal biology. Biomolecules 13(9):1311.
Liegel RP, Michalski MN, Vaidya S, Bitterman E, Finnerty E, Menke CA, Diegel CR, Zhong ZA, Williams BO, Stottman RW. 2023. Successful therapeutic intervention in new mouse models of frizzled 2-associated congenital malformations. Development 150(3).
Craig SEL, Michalski MN, Williams BO. 2023. Got WNTS? Insight into bone health from a WNT perspective. Curr Top Dev Biol152:327–346.
Kitchen-Goosen SM, Schumacher H, Good J, Patterson AL, Boguslawski EA, West RA, Williams BO, Hostetter G, Agnew DW, Teixeira JM, Alberts AS. 2023. Endometrial hyperplasia with loss of APC in a novel population of Lyz2-expressing mouse in endometrial epithelial cells. Carcinogen 44(1):54–64.
2022
Maupin KA, Diegel CR, Stevens PD, Dick D, VAI Vivarium and Transgenics Core, Williams BO. 2022. Mutation of the galectin-3 glycan-binding domain (Lgals3-R200S) enhances cortical bone expansion in male mice and trabecular bone mass in female mice. FEBS Open Bio 12(10):1717–1728.
Hollis K, Williams BO. 2022. Untangling frizzled functions. FASEB J 36(Supplement 1).
Liegel R, Michalski MN, Vaidya S, Bittermann E, Finnerty E, Menske C, Diegel CR, Zhong ZA, Williams BO, Stottmann R. 2022. Dkk1 inhibition normalizes limb phenotypes in a mouse model of Fzd2 associated omodysplasia Robinow syndromes. bioRXiv.
Ubels JL, Lin CM, Antonetti DA, Diaz-Coranguez M, Diegel CR, Williams BO. 2022. Structure and function of the retina of low-density lipoprotein receptor-related protein 5 (Lrp5)-deficient rats . Exp Eye Res 217: 108977.
2021
Zhong ZA, Michalski, MN, Stevens PD, Sall EA, Williams BO. 2021. Regulation of Wnt receptor activity: Implications for therapeutic development in colon cancer. J Biol Chem:100782.
. 2021. Mithramycin induces promoter reprogramming and differentiation of rhabdoid tumor. EMBO Mol Med 13(2):e12640.
Michalski MN, Diegel CR, Zhong ZA, Suino-Powell K, Blazer L, Adams J, VAI Vivarium and Transgenics Core, Beddows I, Melcher K, Sidhu SS, Angers S, Williams BO. 2021. Generation of a new Frizzled 2 flox mouse to clarify its role in development. bioRxiv.
Stevens PD, Williams BO. 2021. LGR4: Not just for Wnt anymore? Cancer Res 81(17):4397–4398.
Foxa GE, Liu Y, Turner LM, Robling AG, Yang T, Williams BO. 2021. Generation and characterization of mouse models for skeletal disease. Methods Mol Biol 2221:165–191.
2020
Diegel CR, Hann S, Ayturk UM, Hu JCW, Lim K, Droscha CJ, Madaj ZB, Foxa GE, Izaguirre I, VAI Vivarium and Transgenics Core, Paracha N, Pidhaynyy B, Dowd TL, Robling AG, Warman ML, Williams BO. 2020. An osteocalcin-deficient mouse strain without endocrine abnormalities. PLOS Genet.
Maupin KA, Dick D, Lee J, Williams BO. 2020. Loss of Lgals3 protects against gonadectomy-induced cortical bone loss in mice. Calcif Tiss Intl.
Lim KE, Bullock WA, Horan DJ, Williams BO, Warman ML, Robling AG. 2020. Co-deletion of Lrp5 and Lrp6 in the skeleton severely diminishes bone gain from sclerostin antibody administration. Bone 143:115708.
Ubels JL, Diegel CR, Foxa GE, Ethen NJ, Lensing JN, Madaj ZB, VAI Vivarium and Transgenics Core, Williams BO. 2020. Low-density lipoprotein-related protein 5 (LRP5)-deficient rats have reduced bone mass and abnormal development of the retinal vasculature. The CRISPR Journal 3(4):284–298.
Jeong W, Kim S, Lee U, Zhong ZA, Savitskiy M, Kwon H, Him J, Lee T, Cho JW, Williams BO, Katanaev VL, Jho EH. 2020. Lrp6 senses the level of nutrients and regulates Hippo signaling. EMBO Rep 21(9):e50103.
Diegel CR, Hann S, Ayturk UM, Hu JCW, Lim K, Droscha CJ, Madaj ZB, Foxa GE, Izaguirre I, VAI Vivarium and Transgenics Core, Robling AG, Warman ML, Williams BO. 2020. Independent validation of experimental results requires timely and unrestricted access to animal models and reagents. PLOS Genetics 16(6):e1008940.
Kushwaha P, Kim S, Foxa GE, Michalski MN, Williams BO, Tomlinson RE, Riddle RC. 2020. Frizzled-4 is required for normal bone acquisition despite compensation by Frizzled-8. J Cell Physiol. 235(10):6673–6683.
Williams BO, Johnson ML. 2020. Wnt signaling and bone cell activity. In: Principles of Bone Biology. Elsevier.
2019
Bergsma A, Ganguly SS, Wiegand ME, Dick D, Williams BO, Miranti CK. 2019. Regulation of cytoskeleton and adhesion in osteoclasts by tetraspanin CD82. Bone Rep. 10:100196.
Mirabelli CK, Nusse R, Tuveson DA, Williams BO. 2019. Perspectives on the role of Wnt biology in cancer. Sci Signal 1:eaay4494.
2018
Bergsma A, Ganguly SS, Dick D, Williams BO, Miranti CK. 2018. Global deletion of tetraspanin CD82 attenuates bone growth and enhances bone marrow adipogenesis. Bone 113:105–113.
Lu D, Li J, Liu H, Foxa GE, Weaver K, Li J, Williams BO, Yang T. 2018. LRP1 suppresses bone resorption in mice by inhibiting the RANKL-stimulated NFκB and p38 pathways during osteoclastogenesis. J Bone Miner Res.
Maupin K, Weaver K, Bergsma A, Christie C, Zhong Z, Yang T, Williams BO. 2018. Enhanced cortical bone expansion in Lgals3-deficient mice during aging. Bone Res 6(7).
Li J, Lu D, Dou H, Liu H, Weaver K, Wang W, Li JD, Yeh E, Williams BO, Zheng L, Yang T. 2018. Demsumoylase SENP6 maintains osteochondroprogenitor homeostasis by suppressing the p53 pathway. Nat Commun 9(1):143.
Madan B, McDonald MJ, Foxa GE, Diegel CR, Williams BO*, Virshup DM*. 2018. Bone loss from Wnt inhibition mitigated by concurrent alendronate therapy. Bone Res 25(6):17.
*Co-corresponding authors
Ganguly SS, Daft PG, Cao J, Meng X, Zhong ZA, Vander Ark A, Meadows A, Madaj Z, Williams BO, Li X. 2018. Loss of myeloid-specific TGF-B signaling decreases CTHRC1 to downregulate bFGF and the development of H1993-induced osteolytic bone lesions. Cancers 10(12):463.
Michalski MN, Williams BO. 2018. A quest for clarity in bone erosion: The role of sequestosome 1 in Paget’s disease of bone. J Biol Chem 293(24):9542–9543.
Nayak G, Odaka Y, Prasad V, Solano AF, Yeo EJ, Vemuraju S, Molkentin JD, Trump A, Williams BO, Rao S, Lang RA. 2018. Developmental vascular regression is regulated by a Wnt/B-catenin, MYC, P21 (CDKN1A) pathway that controls cell proliferation and cell death. Development 145(12):dev154898.
Liu H, Li J, Lu D, Li J, He Y, Williams BO, Yang T. 2018. Ginkgolic acid, a sumoylation inhibitor, promotes adipocyte commitment but suppresses adipocyte terminal differentiation of mouse bone marrow stromal cells. Sci Rep 8(1):2545.
Zhong ZA, Ethen NJ, Williams BO. 2018. Using genetically engineered mouse models to study Wnt signaling in bone development and disease. In: Regulation of Signal Transduction in Human Cell Research, Springer, 119-25.
2017
Valkenburg KC, DeMarzo AM, Williams BO. 2017. Deletion of tumor suppressors adenomatous polyposis coli and Smad4 in murine luminal epithelial cells causes invasive prostate cancer and loss of androgen receptor expression. Oncotarget 8(46):80265–80277.
DeBruine ZJ, Ke J, Harikumar KG, Gu X, Borowsky P, Williams BO, Miller LJ, Xu HE, Melcher K. 2017. Wnt5a promotes Frizzled-4 signalosome assembly by stabilizing cysteine-rich domain dimerization. Genes Dev 31(9):916–926.
Li J, Lu D, Liu H, Williams BO, Overbeek PA, Lee B, Zheng L, Yang T. 2017. Sclt1 deficiency causes cystic kidney by activating ERK and STAT3 signaling. Hum Mol Genet 26(15):2949–2960.
Janda CY, Dang LT, You C, Chang J, de Lau W, Zhong ZA, Yan KS, Marecic O, Siepe D, Li X, Moody JD, Williams BO, Clevers H, Kuo CJ, Piehler J, Baker D, Garcia KC. 2017. Surrogate Wnt ligands that phenocopy canonical Wnt/B-catenin signaling. Nature 545(7653):234–237.
Walls GV, Stevenson M, Lines KE, Newey PJ, Reed AAC, Bowl MR, Jeyabalan J, Harding B, Bradley KJ, Manek S, Chen J, Wang P, Williams BO, Teh BT, Thakker RV. 2017. Mice deleted for Cell division cycle 73 (Cdc73) gene develop parathyroid and uterine tumors: model for the hyperparathyroidism-jaw tumour syndrome. Oncogene.36(28):4025–4036.
Droscha CJ, Diegel CR, Ethen NJ, Burgers TA, McDonald MJ, Maupin KA, Naidu AS, Wang PF, Teh BT, Williams BO. 2017. Osteoblast-specific deletion of Hrpt2/Cdc73 results in high bone mass and increased bone turnover. Bone 98:68–78.
Pridgeon MG, Grohar PJ, Steensma MR, Williams BO. 2017. Wnt signaling in Ewing sarcoma, osteosarcoma, and malignant peripheral nerve sheath tumors. Curr Osteoporos Rep 15(4):239–246.
Yang T, Williams BO. Low-density lipoprotein receptor-related proteins in skeletal development and disease. Physiol Rev 97(3):1211–1228.
Williams BO. 2017. LRP5: From bedside to bench to bone. Bone 102:26–30.
Williams BO, Warman ML. 2017. CRISPR/Cas9 technologies. J Bone Miner Res 32(5):883–888.
2016
Zhong ZA, Ethen NJ, Williams BO. 2016. Use of primary calvarial osteoblasts to evaluate the function of Wnt signaling in osteogenesis. Methods Mol Biol 1481:199–125.
Williams BO. 2016. Genetically engineered mouse models to evaluate the role of Wnt secretion in bone development and homeostasis. Am J Med Genet Semin Med Genet 172C(1):24–26.
Chin AM, Tsai YH, Finkbeiner SR, Nagy NS, Walker EM, Ethen NJ, Williams BO, Battle MA, Spence JR. 2016. A dynamic Wnt/B-catenin signaling environment leads to the Wnt-independent and Wnt-dependent proliferation of embryonic intestinal progenitor cells. Stem Cell Reports 7(5):826–839.
Burgers TA, Vivanco JF, Zahatnansky J, Moren AJ, Mason JJ, Williams BO. 2016. Mice with a heterozygous Lrp6 deletion have impaired fracture healing. Bone Res 4:16025.
Schumacher CA, Joiner DM, Less KD, Drewry MO, Williams BO. 2016. Characterization of genetically engineered mouse models carrying Col2a1-cre-induced deletions of Lrp5 and/or Lrp6. Bone Res 4:15042.
Meng X, Vander Ark A, Lee P, Hostetter G, Bhowmick NA, Matrisian LM, Williams BO, Miranti CK, Li X. 2016. Myeloid-specific TGF-β signaling in bone promotes basic-FGF and breast cancer bone metastasis. Oncogene 35(18):2370–2378.
Weivoda MM, Ruan M, Hachfeld CM, Pederson L, Howe A, Davey R, Zajac J, Kobayashi Y, Williams BO, Westendorf JJ, Khosla S, Oursler MJ. 2016. Wnt signaling inhibits osteoclast differentiation by activating canonical and non-canonical cAMP/PKA pathways. J Bone Miner Res 31(1):65–75.
Rudnicki MA, Williams BO. 2016. Wnt signaling in bone and muscle. Bone 80:60–66.
2015
Oursler MJ, Westendorf JJ, Weivoda MM, Ruan M, Hachfeld CM, Howe A, Davey R, Zajac J, Williams BO, Khosla S. 2015. Response to Wnt signaling pathways. J Bone Miner Res 30(11):2135–2136.
Chen J, Huang D, Rubera I, Futami K, Wang PF, Zickert P, Khoo SK, Dykema KJ, Zhao P, Petillo D, Cao B, Zhang Z, Si S, Schoen SR, Yang XJ, Zhou M, Xiao GQ, Wu G, Nordenskjold M, Tauc M, Williams BO, Furge KA, Teh BT. 2015. Disruption of tubular Flcn expression as a mouse model for renal tumor induction. Kidney Int 88(5):1057–1069.
Zhong ZA, Zahatnansky J, Snider J, Van Wieren E, Diegel CR, Williams BO. 2015. Wntless spatially regulates bone development through β-catenin-dependent and independent mechanisms. Dev Dyn 244(10):1347–1355.
Cheng SL, Ramachandran B, Behrmann A, Shao JS, Mead M, Smith C, Krchma K, Arrendondo YB, Kovacs A, Kapoor K, Brill LM, Perera R, Williams BO, Towler DA. 2015. Vascular smooth muscle LRP6 limits arteriosclerotic calcification in diabetic LDLR–/– mice by restraining noncanonical Wnt signals. Circ Res 117(2):142–156.
Collins CJ, Vivanco JF, Sokn SA, Williams BO, Burgers TA, Ploeg HL. 2015. Fracture healing in mice lacking Pten in osteoblasts: a micro-computed tomography image-based analysis of the mechanical properties of the femur. J Biomech 48(2):310–317.
Peacock JD, Dykema KJ, Toriello HV, Mooney MR, Scholten DJ 2nd, Winn ME, Borgman A, Duesbery NS, Hiemenga JA, Liu C, Campbell S, Nickoloff BP, Williams BO, Steensma MR. 2015. Oculoectodermal syndrome is a mosaic RASopathy associated with KRAS alterations. Am J Med Genet A 167(7):1429–1435.
Qin L, Yin YT, Zheng FJ, Peng LX, Yang CF, Bao YN, Liang YY, Li XJ, Xiang YQ, Sun R, Li AH, Zou RH, Pei XQ, Huang BJ, Kang TB, Liao DF, Zeng YX, Williams BO, Qian CN. 2015. WNT5A promotes stemness characteristics in nasopharyngeal carcinoma cells leading to metastasis and tumorigenesis. Oncotarget 6(12):10239–10252.
Valkenburg KC, Hostetter G, Williams BO. 2015. Concurrent hepsin overexpression and adenomatous polyposis coli deletion causes invasive prostate carcinoma in mice. Prostate 75(14):1579–1585.
Zhong ZA, Peck A, Li S, VanOss J, Snider J, Droscha CJ, Chang TA, Williams BO. 2015. 99mTC-Methylene diphosphonate uptake at injury site correlates with osteoblast differentiation and mineralization during bone healing in mice. Bone Res 3:15013.
2014
Valkenburg KC, Yu X, De Marzo AM, Spiering TJ, Matusik RJ, Williams BO. 2014. Activation of Wnt/b-catenin signaling in a subpopulation of murine prostate luminal epithelial cells induces high grade prostate intraepithelial neoplasia. Prostate 74(15):1506–1520.
Zhong Z, Ethen NJ, Williams BO. 2014. WNT signaling in bone development and homeostasis. Wiley Interdiscip Red Dev Biol 3(6):489–500.
Li C, Williams BO, Cao X, Wan M. 2014. LRP6 in mesenchymal stem cells is required for bone formation during bone growth and bone remodeling. Bone Res 2:14006.
Yang J, Mowry LE, Nejak-Bowen KN, Okabe H, Diegel CR, Lang RA, Williams BO, Monga SP. 2014. b-catenin signaling in murine liver zonation and regeneration: a Wnt-Wnt situation! Hepatology 60(3):964–976.
Scholten DJ 2nd, Timmer CM, Peacock JD, Pelle DW, Williams BO, Steensma MR. 2014. Down regulation of Wnt signaling mitigates hypoxia-induced chemoresistance in human osteosarcoma cells. PLoS One 9(10):e111431.
Cui Y, Niziolek PJ, MacDonald BT, Alenina N, Matthes S, Jacobsen CM, Conlon RA, Brommage R, Powell DR, Bader M, Williams BO, Warman ML, Robling AG. 2014. Reply to Lrp5 regulation of bone mass and gut serotonin synthesis. Nat Med 20(11):1229–1230.
Hoffmann FM, Burgers T, Mason JJ, Williams BO, Sietsema DL, Jones CB. 2014. Biomechanical evaluation of fracture fixation constructs using a variable-angle locked periprosthetic femur plate system. Injury 45(7):1035–1041.
Joiner DM, Less KD, Van Wieren EM, Hess D, Williams BO. 2014. Accelerated and increased joint damage in young mice with global inactivation of mitogen-inducible gene 6 (Mig-6) after ligament and meniscus injury. Arthritis Res Ther 16(2):R81.
Kabiri Z, Greicius G, Madan B, Biechele S, Zhong Z, Zaribafzadeh H, Aliyed J, Wu Y, Bunte RM, Williams BO, Rossant J, Virshup DM. 2014. Stroma provides an intestinal stem cell niche in the absence of epithelial Wnts. Development 141(11):2206–2215.
Lim WH, Liu B, Cheng D, Williams BO, Mah SJ, Helms JA. 2014. Wnt signaling regulates homeostasis of the periodontal ligament. J Periodontal Res 49(6):751–759.
Lim WH, Liu B, Cheng D, Hunter DJ, Zhong Z, Ramos DM, Williams BO, Sharpe PT, Bardet C, Mah SJ, Helms JA. 2014. Wnt signaling regulates pulp volume and dentin thickness. J Bone Miner Res 29(4):892–901.
Staal B, Williams BO, Beier F, Vande Woude GF, Zhang YW. 2014. Cartilage-specific deletion of Mig-6 results in osteoarthritis-like disorder with excessive articular chrondrocyte proliferation. Proc Natl Acad Sci U S A 111(7):2590–2595.
Williams BO. 2014. Insights into the mechanisms of sclerostin action in regulating bone mass accrual. J Bone Min Res 29(1):24–28.
2013
Burgers TA, Williams BO. 2013. Regulation of Wnt/β-catenin signaling within and from osteocytes. Bone 54(2):244–249.
Burgers TA, Hoffmann MF, Collins CJ, Zahatnansky J, Alvarado MA, Morris MR, Sietsma DL, Mason JJ, Jones CB, Ploeg HL, Williams BO. 2013. Mice lacking Pten in osteoblasts have improved intramembranous and late endochondral fracture healing. PLoS One 8(5):e63857.
Hu B, Chen Y, Usmani SZ, Ye S, Qiang W, Papanikolaou X, Heuck CJ, Yaccoby S, Williams BO, Van Rhee F, Barlogie B, Epstein J, Qiang YW. 2013. Characterization of the molecular mechanism of the bone-anabolic activity of carfilzomib in multiple myeloma. PLoS One 8(9):e74191.
Joiner DM, Less KD, Van Wieren EM, Hess D, Williams BO. 2013. Heterozygosity for an inactivating mutation in low-density lipoprotein-related receptor 6 (Lrp6) increases osteoarthritis severity in mice after ligament and meniscus injury. Osteoarthritis Cartilage 21(10):1576–1585.
Joiner DM, Ke J, Zhong Z, Xu HE, Williams BO. 2013. LRP5 and LRP6 in development and disease. Trends Endocrin Metab 24(1):31–39.
Ke J, Xu HE, Williams BO. 2013. Lipid modification in Wnt structure and function. Curr Opin Lipidol 24(2):129–133.
Ke J, Harikumar KG, Jurecky C, Chen C, Gu X, Wang L, Parker N, Cheng Z, Xu W, Williams BO, Melcher K, Miller LJ, Xu HE. 2013. Structure and function of Norrin in assembly and activation of a Frizzled 4–Lrp5/6 complex. Genes Dev 27(21):2305–2319.
Loftus JC, Dhruv H, Tuncali S, Kloss J, Yang Z, Schumacher CA, Cao B, Williams BO, Eschbacher JM, Ross JTD, Tran NL. 2013. TROY (TNFRSF19) promotes glioblastoma cell survival signaling and therapeutic resistance. Mol Cancer Res 11(8): 865–874.
Maupin KA, Droscha CJ, Williams BO. 2013. A comprehensive overview of skeletal phenotypes associated with alterations in Wnt/β-catenin signaling in humans and mice. Bone Res 1(1):27–71.
Riddle RC, Diegel CR, Leslie KM, Van Koevering KK, Faugere MC, Clemens TL, Williams BO. 2013. Lrp5 and Lrp6 exert overlapping functions in osteoblasts during postnatal bone acquisition. PLoS One 8(5):e63323.
Steensma MR, Tyler WK, Shaber AG, Goldring SR, Ross FP, Williams BO, Healey JH, Purdue PE. 2013. Targeting the giant cell tumor stromal cell: functional characterization and a novel therapeutic strategy. PLoS One 8(7):e69101.
Valkenburg KC, Steensma MR, Williams BO, Zhong Z. 2013. Skeletal metastasis: treatments, mouse models, and the Wnt signaling. Chin J Cancer 32(7):380–396.
2012
Fortin SP, Ennis MJ, Schumacher CA, Zylstra-Diegel CR, Williams BO, Ross JTD, Winkles JA, Loftus JC, Symons MH, Tran NL. 2012. Cdc42 and the guanine nucleotide exchange factors Ect2 and Trio mediate Fn14-Rac1-induced migration and invasion of glioblastoma cells. Mol Cancer Res 10(7):958–968.
Ke J, Zhang C, Harikumar KG, Zylstra-Diegel CR, Wang L, Mowry LE, Miller LJ, Williams BO, Xu HE. 2012. Modulation of β-catenin signaling by glucagon receptor activation. PLoS One 7(3):e33676.
Li Y, Ferris A, Lewis BC, Orsulic S, Williams BO, Holland EC, Hughes SH. 2012. The RCAS/TVA somatic gene transfer method in modeling human cancer. In Genetically Engineered Mice for Cancer Research, J.E. Green and T. Ried, eds. Berlin: Springer Verlag, pp. 83–112.
Regard JB, Zhong Z, Williams BO, Yingzi Y. 2012. Wnt signaling in bone development and disease: making stronger bones with Wnts. Cold Spring Harb Perspect Biol4(12):a007997.
Zhong Z, Williams BO. 2012. Integration of cellular adhesion and Wnt signaling: interactions between N-cadherin and LRP5 and their role in regulating bone mass. J Bone Miner Res27(9):1849–1851.
Zhong Z, Williams BO, Steensma MR. 2012. The activation of β-catenin by Gαs contributes to the etiology of phenotypes seen in fibrous dysplasia and McCune-Albright syndrome. IBMS BoneKEy 9:113.
Zhong Z, Baker JJ, Zylstra-Diegel CR, Williams BO. 2012. Lrp5 and Lrp6 play compensatory roles in mouse intestinal development. J Cell Biochem 113(1):31–38.
Zhong Z, Zylstra-Diegel CR, Schumacher CA, Baker JJ, Carpenter AC, Rao S, Yao W, Guan M, Helms JA, Lane NE, Lang RA, Williams BO. 2012. Wntless functions in mature osteoblasts to regulate bone mass. Proc Natl Acad Sci U S A 109(33): E2197–E2204.
2011
Cui Y, Niziolek PJ, MacDonald BT, Zylstra CR, Alenina N, Robinson DR, Zhong Z, Matthes S, Jacobsen CM, Conlon RA, Brommage R, Liu Q, Mseeh F, Powell DR, Yang QM, Zambrowicz B, Gerrits H, Gossen JA, He X, Bader M, Williams BO, Warman ML, Robling AG. 2011. Lrp5 functions in bone to regulate bone mass. Nat Med17(6):684–691.
Joeng KS, Schumacher C, Zylstra-Diegel CR, Long F, Williams BO. 2011. Lrp5 and Lrp6 redundantly control skeletal development in the mouse embryo. Dev Biol 359(2):222–229.
Qian CN, Williams BO. 2011. The induction of high endothelial venule remodeling by primary tumors does not correlate to the metastatic capability of tumor cells. J Blood Lymph 1(1):e102.
Stefater JA 3rd, Lewkowich I, Rao S, Mariggi G, Carpenter AC, Burr AR, Fan J, Ajima R, Molkentin JD, Williams BO, Wills-Karp M, Pollard JW, Yamaguchi T, Ferrara N, Gerhardt H, Lang RA. 2011. Regulation of angiogenesis by a non-canonical Wnt-Flt1 pathway in myeloid cells. Nature 474(7352):511–515.
Valkenburg KC, Williams BO. 2011. Mouse models of prostate cancer. Prostate Cancer 2011: 22 pp.
Wan M, Li J, Herbst K, Zhang J, Yu B, Wu X, Qiu T, Lei W, Lindvall C, Williams BO, Ma H, Zhang F, Cao X. 2011. LRP6 mediates cAMP generation by G protein–coupled receptors through regulating the membrane targeting of Gαs. Sci Signal 4(164):ra15.
2010
Coulibaly MO, Sietsema DL, Burgers TA, Mason J, Williams BO, Jones CB. 2010. Recent advances in the use of serological bone formation markers to monitor callus development and fracture healing. Crit Rev Eukaryot Gene Expr 20(2):105–127.
Diegel CR, Cho KR, El-Naggar AK, Williams BO, Lindvall C. 2010. Mammalian target of rapamycin-dependent acinar cell neoplasia after inactivation of Apc and Pten in the mouse salivary gland: implications for human acinic cell carcinoma. Cancer Res 70(22):9143–9152.
Mason JJ, Williams BO. 2010. SOST and DKK: antagonists of LRP family signaling as targets for treating bone disease. J Osteoporos 2010:460120.
2009
Badders NM, Goel S, Clark RJ, Klos KS, Kim S, Bafico A, Lindvall C, Williams BO, Alexander CM. 2009. The Wnt receptor, Lrp5, is expressed by mouse mammary stem cells and is required to maintain the basal lineage. PLoS One 4(8):e6594.
Castilho RM, Squarize CH, Chodosh LA, Williams BO, Gutkind JS. 2009. mTOR mediates Wnt-induced epidermal stem cell exhaustion and aging. Cell Stem Cell5(3):279–289.
Lindvall C, Zylstra CR, Evans N, West RA, Dykema K, Furge KA, Williams BO. 2009. The Wnt co-receptor Lrp6 is required for normal mouse mammary gland development. PLoS One 4(6):e5813.
Qian CN, Furge KA, Knol J, Huang D, Chen J, Dykema KJ, Kort EJ, Massie A, Khoo SK, Vanden Beldt K, Resau JH, Anema J, Kahnoski RJ, Morreau H, Camparo P, Comperat E, Sibony M, Denoux Y, Molinie V, Vieillefond A, Eng C, Williams BO, Teh BT. 2009. Activation of the PI3K/AKT pathway induces urothelial carcinoma of the renal pelvis: identification in human tumors and confirmation in animal models. Cancer Res 69(21):8256–8264.
Williams BO, Insogna KL. 2009. Where Wnts went: the exploding field of Lrp5 and Lrp6 signaling in bone. J Bone Miner Res 24(2):171–178.
2008
Chen J, Futami K, Petillo D, Peng J, Wang P, Knol J, Li Y, Khoo SK, Huang D, Qian CN, Zhao P, Dykema K, Zhang R, Cao B, Yang XJ, Furge K, Williams BO, Teh BT. 2008. Deficiency of FLCN in mouse kidney led to development of polycystic kidneys and renal neoplasia. PLoS One 3(10):e3581.
Qian Z, Chen L, Fernald AA, Williams BO, Le Beau MM. 2008. A critical role for Apc in hematopoietic stem and progenitor cell survival. J Exp Med 205(9):2163–2175.
Robinson DR, Zylstra CR, Williams BO. 2008. Wnt signaling and prostate cancer. Curr Drug Targets 9(7):571–580.
Shekhar MPV, Gerard B, Pauley RJ, Williams BO, Tait L. 2008. Rad6B is a positive regulator of β-catenin stabilization. Cancer Res 68(6):1741–1750.
VanKoevering KK, Williams BO. 2008. Transgenic mouse strains for conditional gene deletion during skeletal development. IBMS BoneKEy 5(5):151–170.
Wang P, Bowl MR, Bender S, Peng J, Farber L, Chen J, Ali A, Zhang Z, Alberts AS, Thakker RV, Shilatifard A, Williams BO, Teh BT. 2008. Parafibromin, a component of the human PAF complex, regulates growth factors and is required for embryonic development and survival in adult mice. Mol Cell Biol 28(9):2930–2940.
Wolf J, Palmby TR, Gavard J, Williams BO. 2008. Multiple PPPS/TP motifs act in a combinatorial fashion to transduce Wnt signaling through LRP6. FEBS Letters582(2): 255–261.
Zylstra CR, Wan C, VanKoevering KK, Sanders AK, Lindvall C, Clemens TL, Williams BO. 2008. Gene targeting approaches in mice: assessing the roles of LRP5 and LRP6 in osteoblasts. J Musculoskelet Neuronal Interact 8(4):291–293.
2007
Bruxvoort KJ, Charbonneau HM, Giambernardi TA, Goolsby JC, Qian CN, Zylstra CR, Robinson DR, Roy-Burman P, Shaw AK, Buckner-Berghuis BD, Sigler RE, Resau JH, Sullivan R, Bushman W, Williams BO. 2007. Inactivation of Apc in the mouse prostate causes prostate carcinoma. Cancer Res 67(6):2490–2496.
Hinoi T, Akyol A, Theisen BK, Ferguson DO, Greenson JK, Williams BO, Cho KR, Fearon ER. 2007. Mouse model of colonic adenoma-carcinoma progression based on somatic Apc inactivation. Cancer Res 67(20):9721–9730.
Lindvall C, Bu W, Williams BO, Li Y. 2007. Wnt signaling, stem cells, and the cellular origin of breast cancer. Stem Cell Rev 3(2):157–168.
Liu X, Bruxvoort KJ, Zylstra CR, Liu J, Cichowski R, Faugere MC, Bouxsein ML, Wan C, Williams BO, Clemens TL. 2007. Lifelong accumulation of bone in mice lacking Pten in osteoblasts. Proc Natl Acad Sci 104(7):2259–2264.
Wu R, Hendrix-Lucas N, Kuick R, Zhai Y, Schwartz DR, Akyol A, Hanash S, Misek DE, Katabuchi H, Williams BO, Fearon ER, Cho KR. 2007. Mouse model of human ovarian endometroid adenocarcinoma based on somatic defects in the Wnt β-catenin and PI3K/Pten signaling pathways. Cancer Cell 11(4):321–333.
Young JJ, Bromberg-White JL, Zylstra CR, Church JT, Boguslawski E, Resau JH, Williams BO, Duesbery NS. 2007. LRP5 and LRP6 are not required for protective antigen–mediated internalization or lethality of anthrax lethal toxin. PLoS Pathog 3(3):e27.
2006
Inoki K, Ouyang H, Zhu T, Lindvall C, Wang Y, Zhang X, Yang Q, Bennett C, Harada Y, Stankunas K, Wang CY, He X, MacDougald OA, You M, Williams BO, Guan KL. 2006. TSC2 integrates Wnt and energy signals via a coordinated phosphorylation by AMPK and GSK3 to regulate cell growth. Cell 126(5):955–968.
Lindvall C, Evans NC, Zylstra CR, Li Y, Alexander CM, Williams BO. 2006. The Wnt signaling receptor LRP5 is required for mammary ductal stem cell activity and Wnt1-induced tumorigenesis. J Biol Chem 281(46):35081–35087.
Park KS, Jeon SH, Kim SE, Bahk YY, Holmen SL, Williams BO, Chung KC, Surh YJ, Choi KY. 2006. APC inhibits ERK pathway activation and cellular proliferation induced by Ras. J Cell Sci119(5):819–827.
Wang PF, Kong D, VanBrocklin MW, Peng J, Zhang C, Potter SJ, Gao X, Teh BT, Zhang N, Williams BO, Holmen SL. 2006. Simplified method for the construction of gene targeting vectors for conditional gene inactivation in mice. Transgenics 4:215–228.
Zhang C, Kong D, Tan MH, Pappas DL Jr, Wang PF, Chen J, Farber L, Zhang N, Koo HM, Weinreich M, Williams BO, Teh BT. 2006. Parafibromin inhibits cancer cell growth and causes G1 phase arrest. Biochem Biophys Res Commun 350(1):17–24.
2005
Castellone MD, Teramoto H, Williams BO, Druey KM, Gutkind JS. 2005. Prostaglandin E2 promotes colon cancer cell growth through a GS-axin-β-catenin signaling axis. Science 310(5753):1504–1510.
Holmen SL, Williams BO. 2005. Essential role for Ras signaling in glioblastoma maintenance. Cancer Res 65(18) 8250–8255.
Holmen SL, Zylstra CR, Mukherjee A, Sigler R, Faugere MC, Bouxsein M, Deng L, Clemens T, Williams BO. 2005. Essential role of ß-catenin in postnatal bone acquisition. J Biol Chem 280(22):21162–21168.
Holmen SL, Robertson SA, Zylstra CR, Williams BO. 2005. Wnt-independent activation of ß-catenin mediated by a Dkk-1-Frizzled 5 fusion protein. Biochem Biophys Res Commun 328(2):533–539.
Qian CN, Knol J, Igarashi P, Lin F, Zylstra U, Teh BT, Williams BO. 2005. Cystic renal neoplasia following conditional inactivation of Apc in mouse renal tubular epithelium. J Biol Chem280(5):3938–3945.
Ai M, Holmen SL, Van Hul W, Williams BO, Warman ML. 2005. Reduced affinity to and inhibition by DKK1 form a common mechanism by which high bone mass-associated missense mutations in LRP5 affect canonical Wnt signaling. Mol Cell Biol 25(12):4946–4955.
Robertson SA, Schoumans J, Looyenga BD, Yuhas JA, Zylstra CR, Koeman JM, Swiatek PJ, Teh BT, Williams BO. 2005. Spectral karyotyping of sarcomas and fibroblasts derived from Ink4a/Arf-deficient mice reveals chromosomal instability in vitro. Int J Oncol 26(3):629–634.
Wang PF, Kong D, VanBrocklin MW, Peng J, Zhang C, Potter SJ, Gao X, Teh BT, Zhang N, Williams BO, Holmen SL. 2005. Simplified method for the construction of gene targeting vectors for conditional gene inactivation in mice. Transgenics 4:215–218.
2004
Bromberg-White JL, Webb CP, Patacsil VS, Miranti CK, Williams BO, Holmen SL. 2004. Delivery of short hairpin RNA sequences by using a replication-competent avian retroviral vector. J Virol 78(9):4914–4916.
Holmen SL, Giambernardi TA, Zylstra CR, Buckner-Berghuis BD, Resau JH, Hess JF, Glatt V, Bouxsein ML, Ai M, Warman ML, Williams BO. 2004. Decreased BMD and limb deformities in mice carrying mutations in both Lrp5 and Lrp6. J Bone Miner Res 19(12): 2033–2040.
Spike BT, Dirlam A, Dibling BC, Marvin J, Williams BO, Jacks T, Macleod KF. 2004. The Rb tumor suppressor is required for stress erythropoiesis. EMBO J 23(21): 4319–4329.
2002
Holmen SL, Salic A, Zylstra CR, Kirschner MW, Williams BO. 2002. A novel set of Wnt-Frizzled fusion proteins identifies receptor components that activate beta -catenin-dependent signaling. J Biol Chem 277(38):34727–34735.
Takahashi M, Yang XJ, Lavery TT, Furge KA, Williams BO, Tretiakova M, Montag A, Vogelzang NJ, Re GG, Garvin AJ, Soderhall S, Kagawa S, Hazel-Martin D, Nordenskjold A, Teh BT. 2002. Gene expression profiling of favorable histology Wilms tumors and its correlation with clinical features. Cancer Res 62(22): 6598–6605.
Yang H, Williams BO, Hinds PW, Shih TS, Jacks T, Bronson RT, Livingston DM. 2002. Tumor suppression by a severely truncated species of retinoblastoma protein. Mol Cell Biol 22(9):3103–3110.
2001
Lipinski MM, Macleod KF, Williams BO, Mullaney TL, Crowley D, Jacks T. 2001. Cell-autonomous and non-cell-autonomous functions of the Rb tumor suppressor in developing central nervous system. EMBO J 20(13):3402–3413.
2000
Dunn KJ, Williams BO, Li Y, Pavan WJ. 2000. Neural crest-directed gene transfer demonstrates Wnt1 role in melanocyte expansion and differentiation during mouse development. Proc Natl Acad Sci U S A 97(18):10050–10055.
Sage J, Mulligan GJ, Attardi LD, Miller A, Chen S, Williams BO, Theodorou E, Jacks T. 2000. Targeted disruption of the three Rb-related genes leads to loss of G(1) control and immortalization. Genes Dev 14(23):3037–3050.
Williams BO, Barish GD, Klymkowsky MW, Varmus HE. 2000. A comparative evaluation of beta-catenin and plakoglobin signaling activity. Oncogene 19(50): 5720–5728.
1999
Klymkowsky MW, Williams BO, Barish GD, Varmus HE, Vourgourakis YE. 1999. Membrane-anchored plakoglobins have multiple mechanisms of action in Wnt signaling. Mol Biol Cell 10(10):3151–3169.
Zorn AM, Barish GD, Williams BO, Lavender P, Klymkowsky MW, Varmus HE. 1999. Regulation of Wnt signaling by Sox proteins: XSox17 alpha/beta and XSox3 physically interact with beta-catenin. Mol Cell 4(4):487–498.
1998
Yamasaki L, Bronson R, Williams BO, Dyson NJ, Harlow E, Jacks T. 1998. Loss of E2F-1 reduces tumorigenesis and extends the lifespan of Rb1(+/-)mice. Nat Genet 18(4):360–364.
1996
Herrera RE, Sah VP, Williams BO, Makela TP, Weinberg RA, Jacks T. 1996. Altered cell cycle kinetics, gene expression, and G1 restriction point regulation in Rb-deficient fibroblasts. Mol Cell Biol 16(5):2402–2407.
Lee MH, Williams BO, Mulligan G, Mukai S, Bronson RT, Dyson N, Harlow E, Jacks T. 1996. Targeted disruption of p107: functional overlap between p107 and Rb. Genes Dev 10(13):1621–1632.
1995
Sah VP, Attardi LD, Mulligan GJ, Williams BO, Bronson RT, Jacks T. 1995. A subset of p53-deficient embryos exhibit exencephaly. Nat Genet 10(2):175–180.
Tischler AS, Shih TS, Williams BO, Jacks T. 1995. Characterization of pheochromocytomas in a mouse strain with a targeted disruptive mutation of the neurofibromatosis gene Nf1. Endocr Pathol 6(4):323–335.
Haas AL, Baboshina O, Williams BO, Schwartz LM. 1995. Exponential increases in ubiquitin conjugate pools accompany the developmentally programmed cell death of insect skeletal muscle. J Biol Chem 270(16):9407–9412.
1994
Jacks T, Remington L, Williams BO, Schmitt EM, Halachmi S, Bronson RT, Weinberg RA. 1994. Tumor spectrum analysis in p53-mutant mice. Curr Biol 4(1):1–7.
Morgenbesser SD, Williams BO, Jacks T, DePinho RA. 1994. p53-dependent apoptosis produced by Rb-deficiency in the developing mouse lens. Nature 371(6492):72–74.
Slebos RJ, Lee MH, Plunkett BS, Kessis TD, Williams BO, Jacks T, Hedrick L, Kastan MB, Cho KR. 1994. p53-dependent G1 arrest involves pRB-related proteins and is disrupted by the human papillomavirus 16 E7 oncoprotein. Proc Natl Acad Sci U S A 91(12):5320–5324.
Williams BO, Morgenbesser SD, DePinho RA, Jacks T. 1994. Tumorigenic and developmental effects of combined germ-line mutations in Rb and p53. Cold Spring Harb Symp Quant Biol 59: 449–457.
Williams BO, Remington L, Albert DM, Mukai S, Bronson RT, Jacks T. 1994. Cooperative tumorigenic effects of germline mutations in Rb and p53. Nat Genet 7(4):480–484.
Williams BO, Schmitt EM, Remington L, Bronson RT, Albert DM, Weinberg RM, Jacks T. 1994. Extensive contribution of Rb-deficient cells to adult chimeric mice with limited histopathological consequences. EMBO J 13(18):4251–4259.
1993
Chen J, Gorman JR, Stewart V, Williams BO, Jacks T, Alt FW. 1993. Generation of normal lymphocyte populations by Rb-deficient embryonic stem cells. Curr Biol 3(7):405–413.
Zettergren LD, Cutlan R, Williams BO. 1991. Embryonic dorsal region compartments in B lymphopoiesis: Rana pipiens, Gallus gallus, and Mus musculus. Dev Comp Immunol 15:105S.
Our research projects investigate how Wnt signaling shapes development and disease through diverse but interconnected mechanisms. By defining how cells coordinate their behavior during tissue formation, we can uncover how Wnt pathways are co-opted in human disease. Each of our projects addresses a fundamental question in Wnt biology. Together, these efforts reflect our lab’s commitment to connecting molecular mechanisms with physiological function and disease.
To study Wnt signaling in vivo and in disease-relevant contexts, we employ a wide-ranging set of experimental systems and technologies. These include mouse genetics, organoid and tissue culture models, and advanced spatial profiling approaches such as spatial transcriptomics. By integrating these tools, we can examine how Wnt signaling operates across scales, from receptor-level mechanisms to tissue organization, providing a comprehensive view of how Wnt pathways shape development and disease.
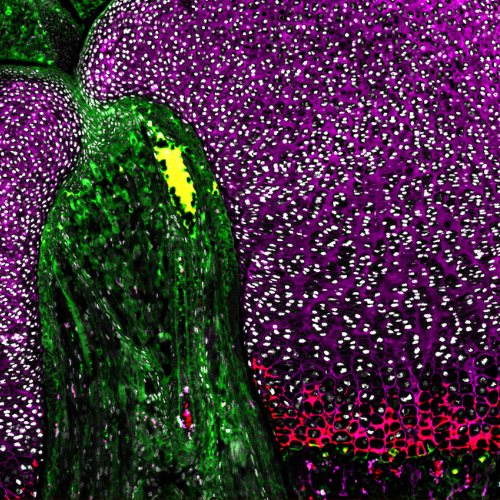
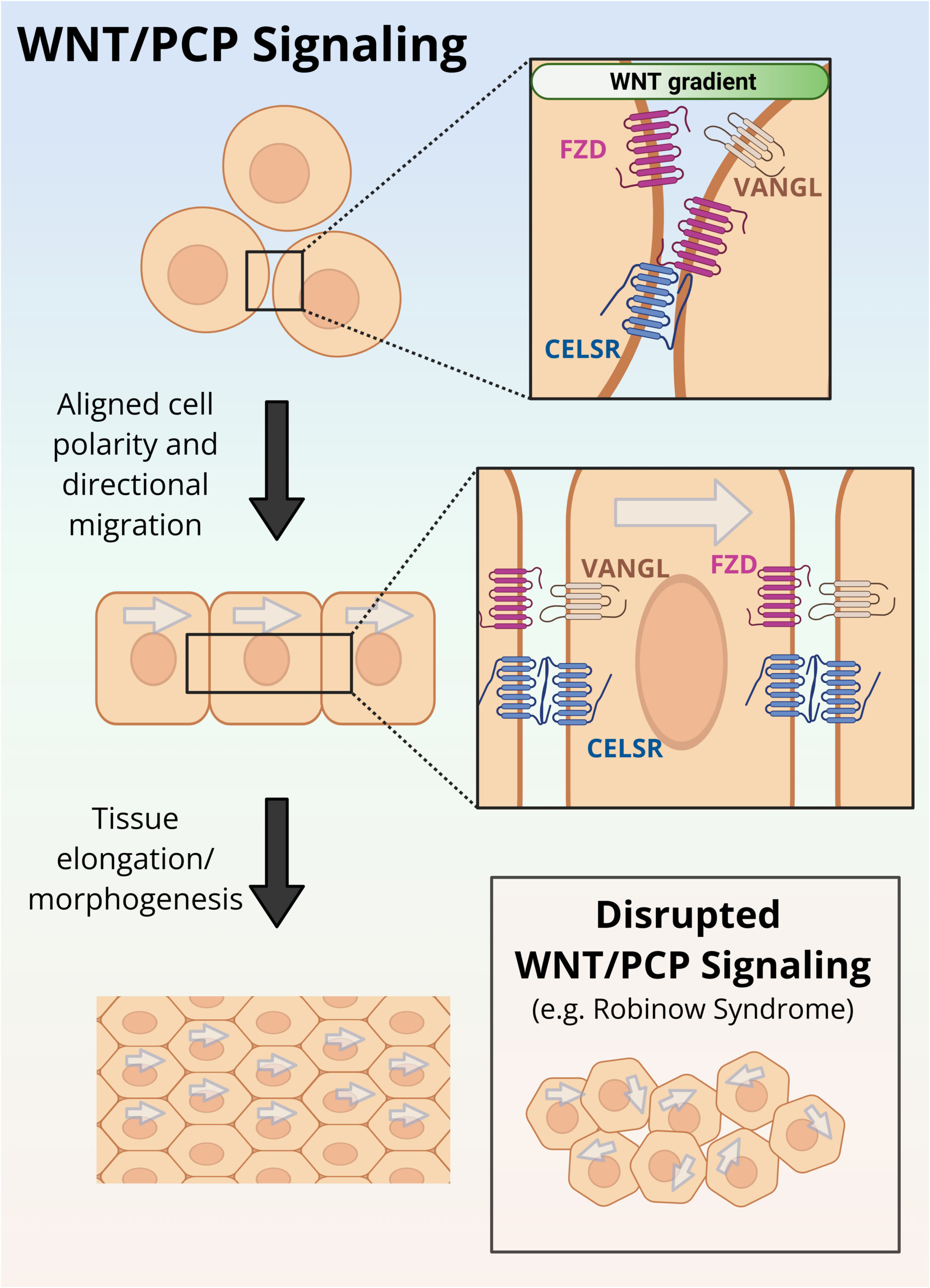
How does noncanonical Wnt signaling coordinate tissue organization and planar cell polarity?
Noncanonical Wnt signaling pathways, including planar cell polarity (PCP) signaling, are essential for tissue organization, morphogenesis and coordinated cell behaviors such as directional migration and oriented cell division. Disruption of these pathways leads to profound developmental defects and underlies human disorders, including Robinow syndrome.
Our lab investigates how noncanonical Wnt ligands and Frizzled receptors regulate PCP signaling during development, with a focus on how signaling is coordinated across tissues to drive morphogenetic movements. Using genetic models, we dissect how mutations in PCP components alter tissue architecture, cell polarity and developmental outcomes. By linking molecular signaling events to tissue-scale organization, we aim to understand how noncanonical Wnt pathways ensure robust patterning during development and how their disruption leads to disease.
Image caption: Created with BioRender.com
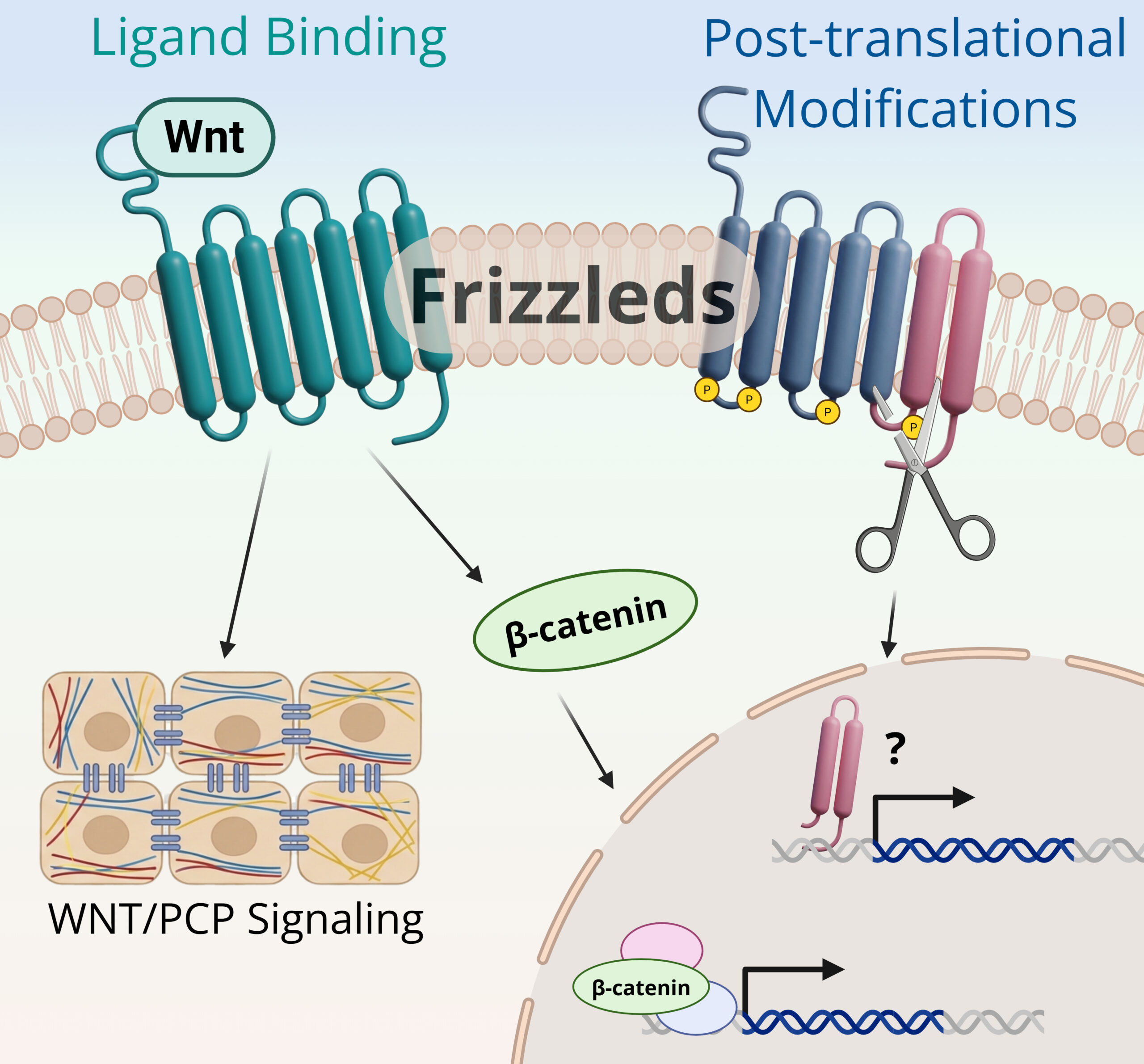
How do Frizzled receptors regulate Wnt signaling specificity and pathway output?
Frizzled receptors are central hubs in Wnt signaling, yet the mechanisms that regulate their signaling specificity and activity in vivo remain poorly defined. Individual Frizzled receptors can participate in both canonical and noncanonical pathways, indicating that receptor regulation plays a critical role in determining pathway output.
Our work focuses on the molecular regulation of Frizzled receptors, including proteolytic processing, receptor trafficking and signaling crosstalk. We are particularly interested in how receptor cleavage and post-translational modifications influence downstream signaling decisions, and how distinct receptor states contribute to context-dependent Wnt responses. By defining how Frizzled receptors integrate extracellular cues and intracellular regulatory mechanisms, we aim to uncover new principles of Wnt signal diversification.
Image caption: Created with BioRender.com
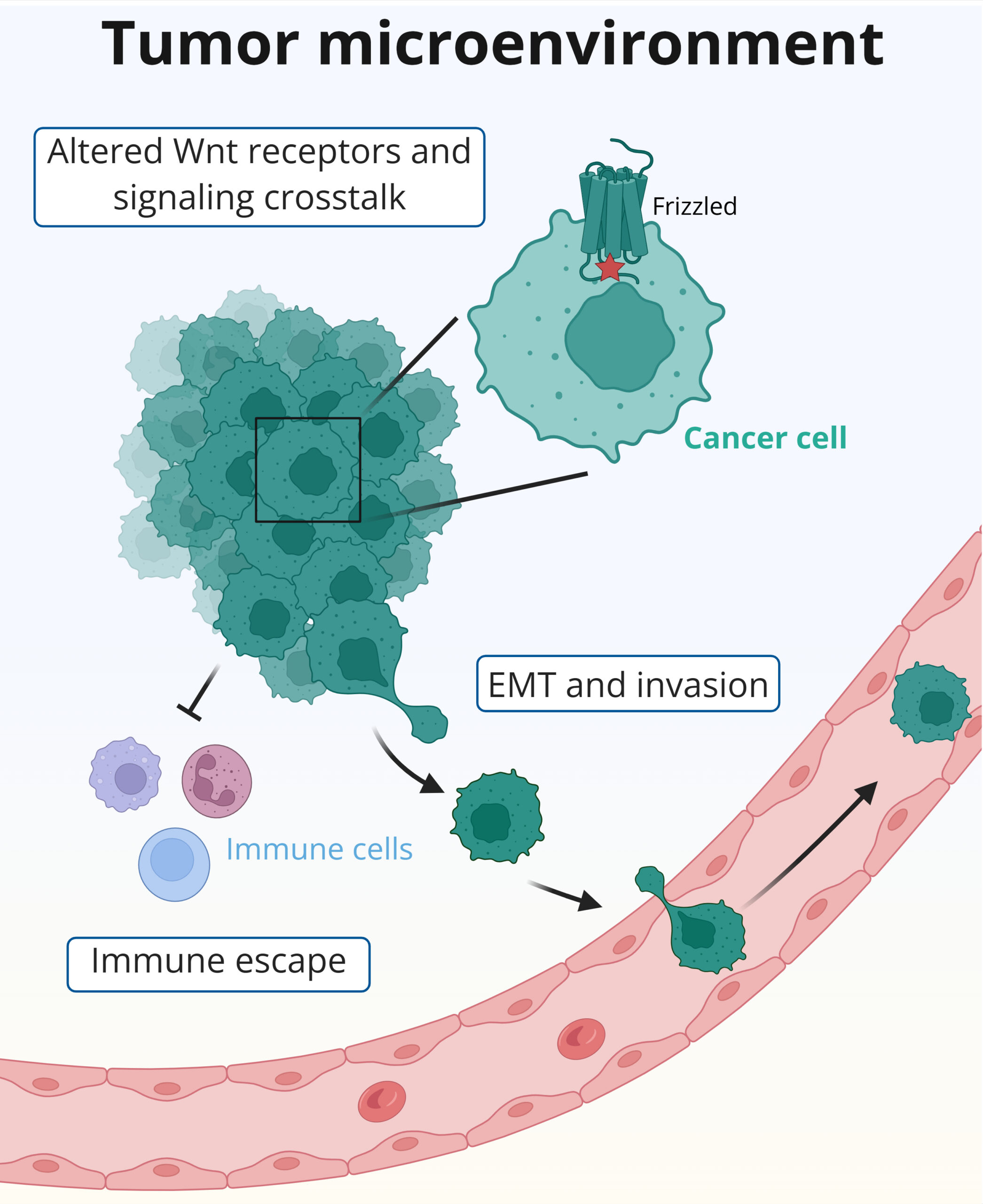
How does altered Wnt signaling drive tumor progression and cellular plasticity in cancer?
Aberrant Wnt signaling is a hallmark of many cancers, where it promotes tumor growth, cellular plasticity and resistance to therapy. While mutations in downstream pathway components are well studied, less is known about how receptor-level alterations and noncanonical Wnt signaling contribute to tumor progression.
Our cancer-focused research examines how altered Wnt signaling drives tumor progression, epithelial-to-mesenchymal transition (EMT) and interactions with the tumor microenvironment. We are particularly interested in pancreatic ductal adenocarcinoma (PDAC), an extremely aggressive cancer with limited therapeutic options. Using genetically engineered mouse models and organoid systems, we investigate how Wnt receptor regulation and pathway crosstalk shape tumor behavior and immune evasion.
Image caption: Created with BioRender.com


Cassandra Diegel, B.S.
Senior Lab Manager, Department of Cell Biology

Megan Michalski, D.D.S., Ph.D.
Research Scientist, Department of Cell Biology

Michelle Minard, B.S.
Senior Administrative Assistant II, Core Technologies and Services

Cayden Steinebach
Student Intern, Williams Laboratory

Alex Zhong, Ph.D.
Research Scientist, Department of Cell Biology

